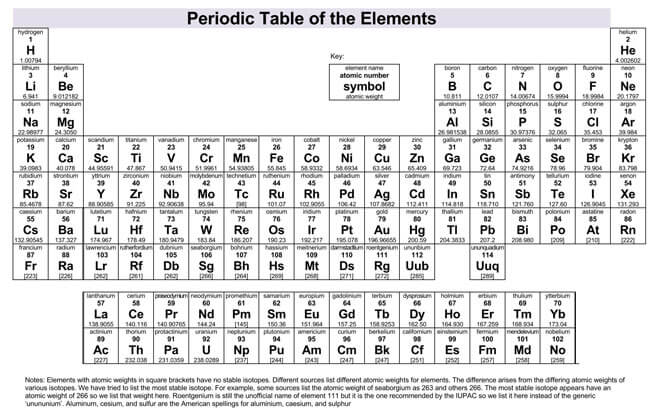
 The periodic table is used by chemists and other scientists as a comprehensive reference source. The metallic characteristics of the elements increase diagonally from right to left. They’re ductile, malleable, and, with the exception of mercury, solid. Metallic Characteristics – Metals are good conductors of heat and electricity.The strength of an atom’s electronegativity increases among the elements as you move from left to right across a period. Electronegativity – Electronegativity is how well an atom can attract a pair of bonding electrons in molecules.Electron affinity increases from left to right across the periodic table. Electron Affinity – The opposite of ionisation energy, electron affinity is the quantity of energy that’s released as an electron is added to an atom.The ionisation energy increases as you move from left to right and decreases from top to bottom of the periodic table. 
Ionisation Energy – This is the energy needed to remove an electron from the outermost orbital of a gaseous atom or ion.It increases as you go from top to bottom. The atomic radius tends to decrease as you move from left to right across the table. Atomic Radius – You can determine the radius of an atom by measuring the distance between two nuclei of an ionic bond. :max_bytes(150000):strip_icc()/PeriodicTablenames-58b5d9193df78cdcd8d02c4c.jpg)
Here are some properties and trends of chemical elements as listed on the periodic table: Trends are immediately obvious if you understand the way the periodic table is arranged. The periodic table arranges all known elements into periods and groups that correspond to specific chemical properties.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
